
We believe these complementary high-performance modes offer excellent potential for rational design of novel amphipathic alpha-helical biologically active peptides. Ch5c Proteins and Nucleic Acids 5251.notebook 5 Secondary Structure the result of H bonds between the repeating constituents of the polypeptide backbone (NOT the amino acid side chains). Hydrophobic molecules are nonpolar and uncharged and,as a conse-quence, are relatively insoluble in water.
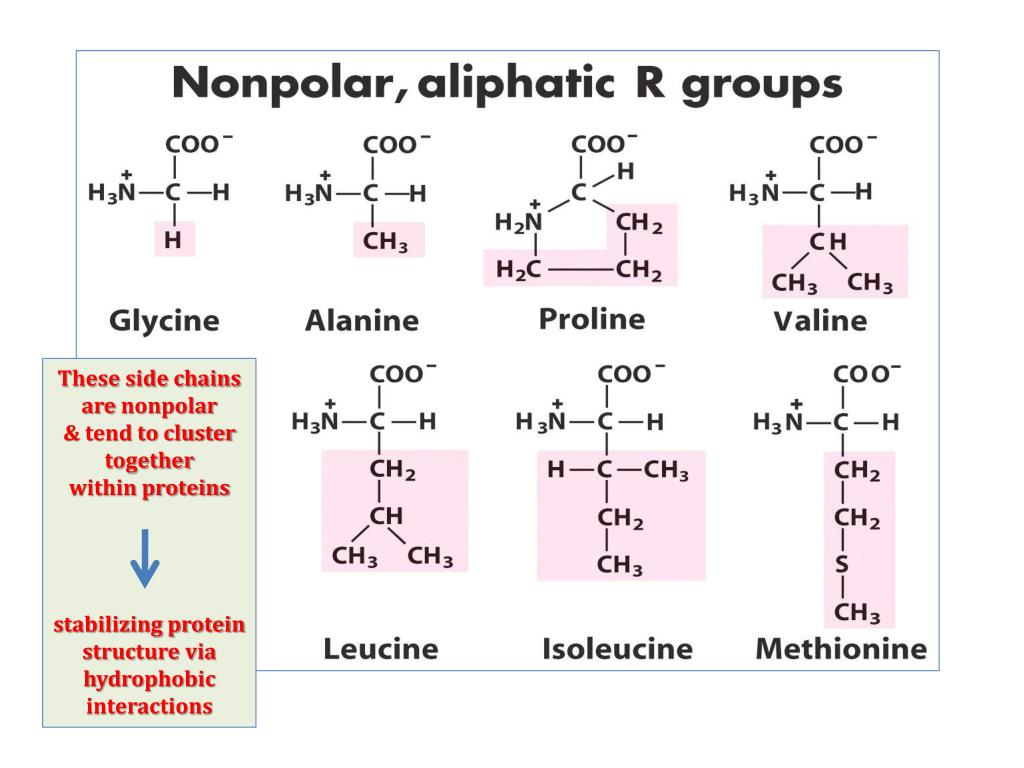
Further, RP-HPLC and HILIC/CEX were demonstrated to be excellent monitors of hydrophilicity/hydrophobicity variations where amino acid substitutions were made in these respective faces. In polymers, hydrophilic side chains tend to associate with other hydrophilic side chains, or with water molecules, usually by means of hydrogen bonds. Our results clearly show that RP-HPLC and HILIC/CEX are best suited for resolving amphipathic peptides where substitutions are made in the non-polar and polar faces, respectively. The present study reports our initial results of employing the complementary separation modes of reversed-phase high-performance liquid chromatography (RP-HPLC) and hydrophilic interaction/cation-exchange chromatography (HILIC/CEX) to monitor the effect on apparent peptide hydrophilicity/hydrophobicity and amphipathicity of substituting single L- or D-amino acids into the centre of the non-polar or polar faces of a 26-residue biologically active amphipathic alpha-helical peptide, V681. Here we are interested in studying the hydrophobic. These different 20 amino acid residues have been grouped into three major classes: hydrophobic (F, M, W, I, V, L, P, A), hydrophilic (N, C, Q, G, S, T, Y), and charged (R, D, E, H, K). The ability to monitor precisely the hydrophobicity/hydrophilicity effects of amino acid substitutions in both the non-polar and polar faces of amphipathic alpha-helical peptides is critical in such areas as the rational de novo design of more effective antimicrobial peptides. Moreover, each of the 20 amino acids has a different side chain and different physicochemical properties.

